EPA Temporarily Amends Process for Active Ingredient Approval to Boost Supply of Disinfectants During COVID-19 Pandemic
To help ensure the availability of disinfectants during the COVID-19 pandemic, the U.S. Environmental Protection Agency (EPA) announced on Monday it was temporarily amending PRN 98-10 to allow manufacturers of certain registered EPA disinfectants to obtain certain “commodity” active ingredients from any source of suppliers without checking with the agency first. EPA announced a similar action on certain inert ingredients last week.
Under the temporary policy, registrants of currently registered pesticide disinfectant products on EPA’s List N: Disinfectants for Use Against SARS-CoV-2 may use any source of the active ingredients listed below without first having to apply for and receive EPA approval of an amendment to their pesticide registration. The temporary policy applies to the following list of active ingredients, provided that the resulting formulation is chemically similar to the current source (i.e., meets the criteria given in Section III.A. of PR Notice 98-10):
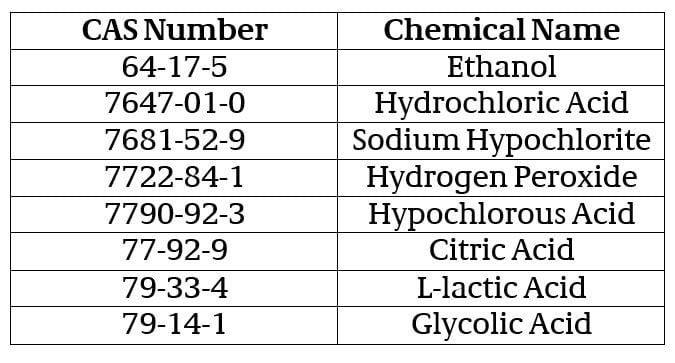
Registrants who wish to take advantage of this temporary policy must first submit an application via the CDX Portal along with a cover letter to EPA that includes the following information:
- In the subject line, state: “Notification per TEMPORARY AMENDMENT TO PR NOTICE 98-10 for EPA Registration No. XXXXXX and [insert product name];”
- Include the active ingredient for which the source is changing and
- The following statement:
“[Name of Registrant] is notifying EPA of its intent to use one or more alternate, unregistered sources of active ingredient listed in the TEMPORARY AMENDMENT TO PESTICIDE REGISTRATION (PR) NOTICE 98-10 (Insert date or other citation) in the formulation of EPA Registration No. [xxx-xx, etc.]. Each source is chemically identical to (i.e., within the certified limits of) the active ingredients in the Confidential Statements of Formula previously accepted by EPA [insert CSF date(s)]. This self-certification is consistent with the provisions of PR Notice 98-10 and no other changes have been made to the confidential statement of formula or labeling of this product. Further, I confirm that the ingredients statement of this label remains truthful. I understand that it is a violation of 18 U.S.C. Sec. 1001 to willfully make any false statement to EPA. I further understand that if this self-certification is not consistent with the terms of PR Notice 98-10 and 40 CFR 152. 46, this product may be in violation of FIFRA and I may be subject to enforcement actions and penalties under section 12 and 14 of FIFRA.”
Registrants may begin to distribute or sell a product modified under the temporary policy once EPA receives the notification.
For more information on EPA’s temporary policy, please contact Chase Dressman or Kimberly DalSanto.
Please visit our COVID-19 Toolkit for all of Taft’s updates on the coronavirus.
In This Article
You May Also Like
EPA Withdraws Endangerment Finding, Rescinds Vehicle GHG Emission Standards EPA Tightens NOx Standards for New Stationary Combustion Turbines